 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Victan, Meilax, Ronlax |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | Hepatic |
| Elimination half-life | 51-103 h |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.044.976 |
| Chemical and physical data | |
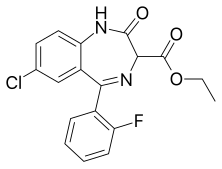
| Formula | C18H14ClFN2O3 |
| Molar mass | 360.77 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Ethyl loflazepate[2] (marketed under the brand names Meilax, Ronlax and Victan)[3][4][5] is a drug which is a benzodiazepine derivative. It possesses anxiolytic, anticonvulsant, sedative and skeletal muscle relaxant properties.[6] In animal studies it was found to have low toxicity, although in rats evidence of pulmonary phospholipidosis occurred with pulmonary foam cells developing with long-term use of very high doses.[7] Its elimination half-life is 51–103 hours.[8] Its mechanism of action is similar to other benzodiazepines. Ethyl loflazepate also produces an active metabolite which is stronger than the parent compound.[9] Ethyl loflazepate was designed to be a prodrug for descarboxyloflazepate, its active metabolite. It is the active metabolite which is responsible for most of the pharmacological effects rather than ethyl loflazepate.[10] The main metabolites of ethyl loflazepate are descarbethoxyloflazepate, loflazepate and 3-hydroxydescarbethoxyloflazepate.[11] Accumulation of the active metabolites of ethyl loflazepate are not affected by those with kidney failure or impairment.[12] The symptoms of an overdose of ethyl loflazepate include sleepiness, agitation and ataxia. Hypotonia may also occur in severe cases. These symptoms occur much more frequently and severely in children.[13] Death from therapeutic maintenance doses of ethyl loflazepate taken for 2 – 3 weeks has been reported in 3 elderly patients. The cause of death was asphyxia due to benzodiazepine toxicity.[14] High doses of the antidepressant fluvoxamine may potentiate the adverse effects of ethyl loflazepate.[15]
Ethyl loflazeplate is commercialized in Mexico, under the trade name Victan. It is officially approved for the following conditions:[16][17]
- Anxiety
- Post-trauma anxiety
- Anxiety associated with severe neuropathic pain
- Generalized anxiety disorder (GAD)
- Obsessive–compulsive disorder
- Panic attack
- Delirium tremens