 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Restas |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral, Intravenous |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 80-90% |
| Metabolism | Hepatic |
| Elimination half-life | 60-90 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
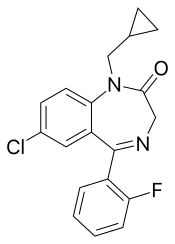
| Formula | C19H16ClFN2O |
| Molar mass | 342.80 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Flutoprazepam (Restas) is a drug which is a benzodiazepine. It was patented in Japan by Sumitomo in 1972[1] and its medical use remains mostly confined to that country. Its muscle relaxant properties are approximately equivalent to those of diazepam - however, it has more powerful sedative, hypnotic, anxiolytic and anticonvulsant effects and is around four times more potent by weight compared to diazepam.[2] It is longer acting than diazepam due to its long-acting active metabolites,[3] which contribute significantly to its effects.[4] Its principal active metabolite is n-desalkylflurazepam, also known as norflurazepam, which is also a principal metabolite of flurazepam (trade name Dalmadorm/Dalmane).[5][6]
Flutoprazepam is typically used for the treatment of severe insomnia and may also be used for treating stomach ulcers.[7]
Flutoprazepam does not fall under the international Convention on Psychotropic Substances of 1971, and is currently unscheduled in the United States.[8]
- In Singapore, flutoprazepam is a Class C-Schedule II drug under the Misuse of Drugs Act.
- In Thailand, flutoprazepam is a Schedule III psychotropic substance.
- In Hong Kong, flutoprazepam is regulated under Schedule 1 of Hong Kong's Chapter 134 Dangerous Drugs Ordinance. Flutoprazepam can only be used legally by health professionals and for university research purposes. The substance can be given by pharmacists under a prescription. Anyone who supplies the substance without prescription can be fined $10000 (HKD). The penalty for trafficking or manufacturing the substance is a $5,000,000 (HKD) fine and life imprisonment. Possession of the substance for consumption without license from the Department of Health is illegal with a $1,000,000 (HKD) fine and/or 7 years of jail time.[9]