| |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C12H17NO |
| Molar mass | 191.274 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
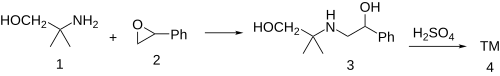
G-130 (GP-130, 2-Phenyl-5,5-dimethyltetrahydro-1,4-oxazine)[1] is a drug with stimulant and anorectic effects, related to phenmetrazine.[2][3][4]