 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.189.975 |
| Chemical and physical data | |
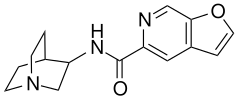
| Formula | C15H17N3O2 |
| Molar mass | 271.314 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
PHA-543,613 is a drug that acts as a potent and selective agonist for the α7 subtype of neural nicotinic acetylcholine receptors, with a high level of brain penetration and good oral bioavailability. It is under development as a possible treatment for cognitive deficits in schizophrenia.[1] It reduces excitotoxicity[2] and protects striatal dopaminergic neurons in rat models.[3] It also potentiates cognitive enhancement from memantine,[4][5] decreases dynorphin release[6] and inhibits GSK-B3.[7]