 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
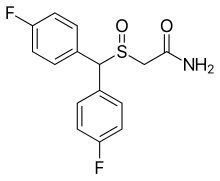
| Formula | C15H13F2NO2S |
| Molar mass | 309.33 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
CRL-40,940 (also known as flmodafinil, bisfluoromodafinil and lauflumide) is the bisfluoro analog of modafinil.[1][2][3][4][5] It is a eugeroic as well as a weak dopamine reuptake inhibitor. Its inventors claim that it is more effective than modafinil and adrafinil, with fewer side effects. It was patented in 2013, and pre-clinical trials have been underway since December 2015.[6]