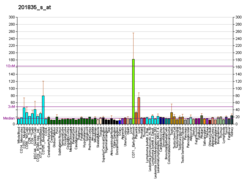
5'-AMP-activated protein kinase subunit beta-1 is an enzyme that in humans is encoded by the PRKAB1 gene.[5][6]
The protein encoded by this gene is a regulatory subunit of the AMP-activated protein kinase (AMPK). AMPK is a heterotrimer consisting of an alpha catalytic subunit, and non-catalytic beta and gamma subunits. AMPK is an important energy-sensing enzyme that monitors cellular energy status. In response to cellular metabolic stresses, AMPK is activated, and thus phosphorylates and inactivates acetyl-CoA carboxylase (ACC) and beta-hydroxy beta-methylglutaryl-CoA reductase (HMGCR), key enzymes involved in regulating de novo biosynthesis of fatty acid and cholesterol. This subunit may be a positive regulator of AMPK activity. The myristoylation and phosphorylation of this subunit have been shown to affect the enzyme activity and cellular localization of AMPK. This subunit may also serve as an adaptor molecule mediating the association of the AMPK complex.[6]